Intervention Recording System (IRS) for Pharma
Intervention Recording System (IRS)

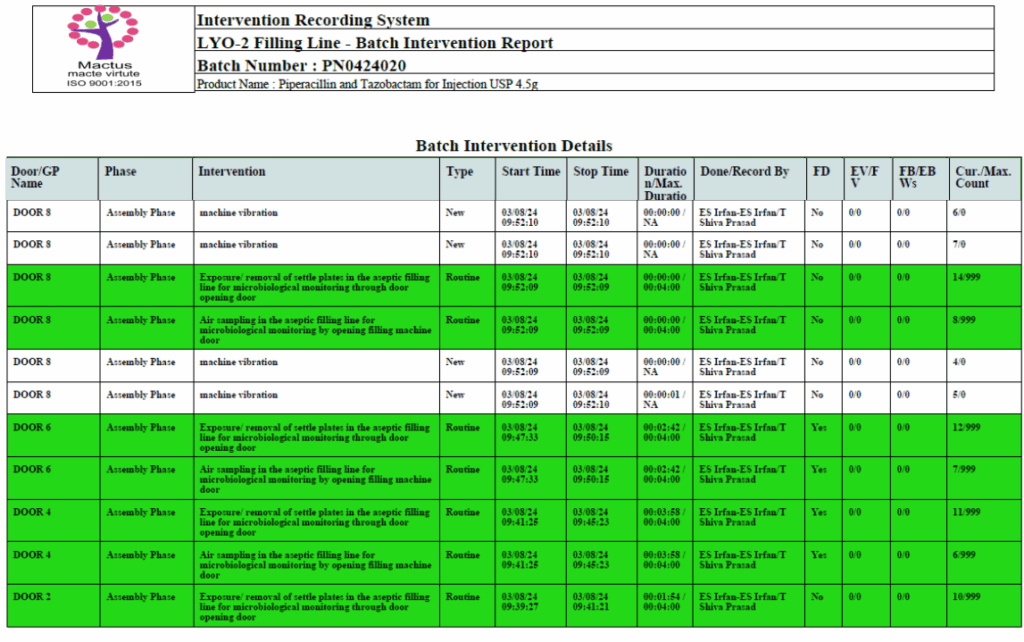
Intervention Recording System (IRS) is a fully configurable digital solution that defines, tracks, manages, and reports all compliance-related activities and interventions performed by operators during fill-finish operations. It enhances adherence to GMP, FDA 21 CFR Part 11, and other regulatory standards through real-time event logging and secure data storage, ensuring audit-ready documentation.
Key Features of IRS

Records interventions by each operator/supervisor with minimum effort
Integrates seamlessly with existing filling machine sensors like doors and glove ports
Tracks each intervention count and duration and requires approval process to perform any intervention, if it exceeds the maximum permitted count
Operators will be guided through text and audio messages based on firms operating procedures and adhering to compliance
The data generated can be processed into intelligent reports, enabling data trending and informed decision-making.
Meets 21 CFR part 11 requirements